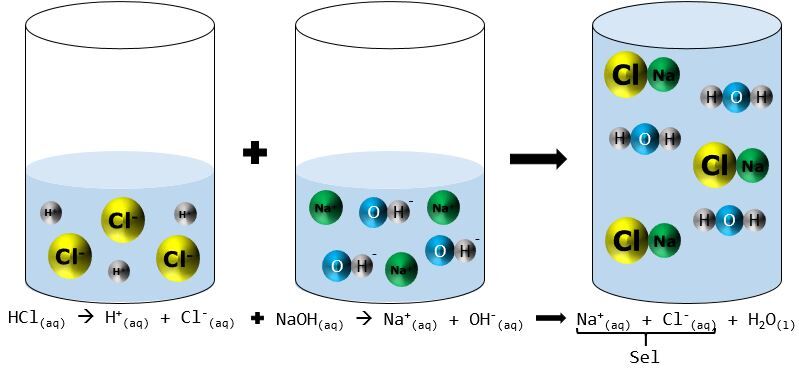
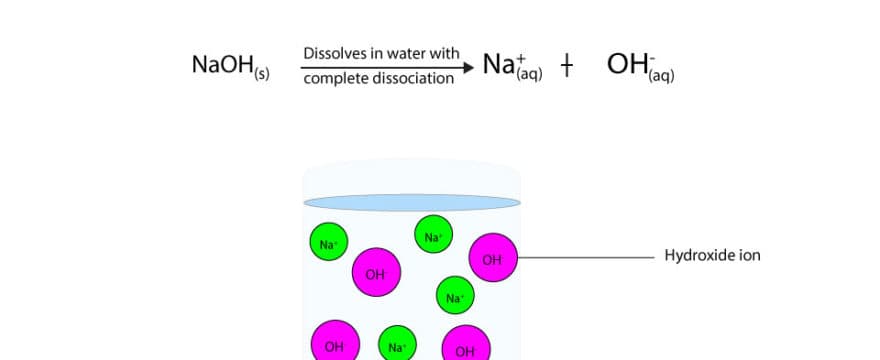
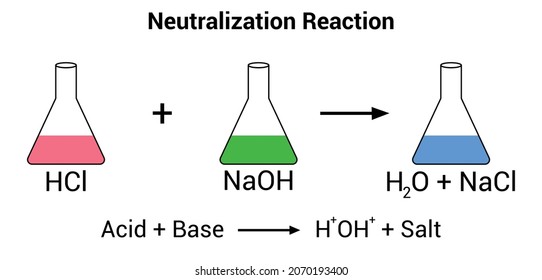
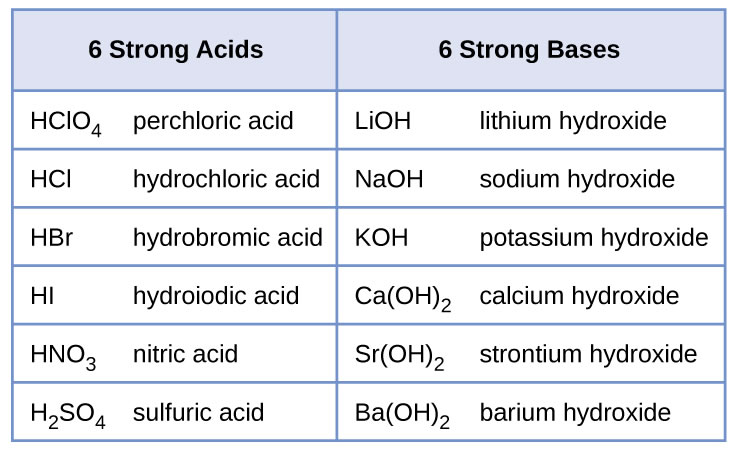
Sodium hydroxide (NaOH) is classified as a strong base. For every mole of sodium hydroxide added to a large volume of water, one mole of what ion enters the solution? | Socratic

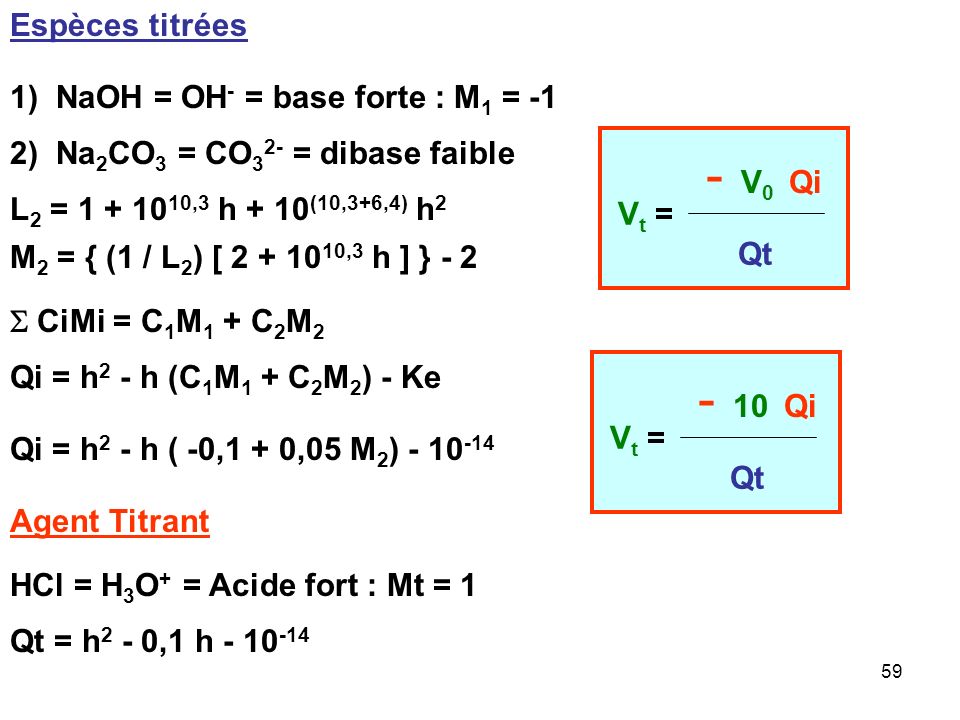
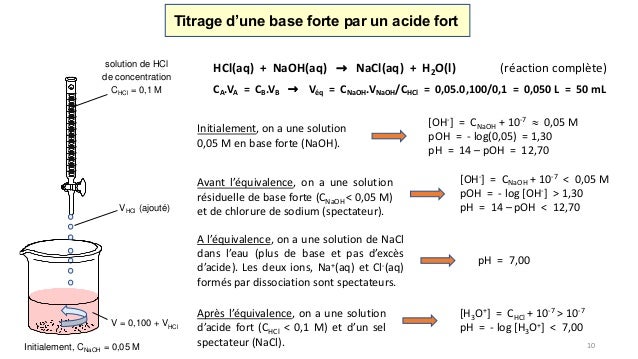
Chine La concentration industrielle Acid-Base en ligne (HNO3, H2SO4, HCl, NAOH, KOH, NACL) mètre pour le traitement de l'eau (ABC-6850) – Acheter Sonde de capteur d'acide sur fr.made-in-china.com

Selective focus of sodium hydroxide base and sulfuric acid solution in brown glass and plastic bottle inside a chemistry laboratory. White background Stock Photo - Alamy

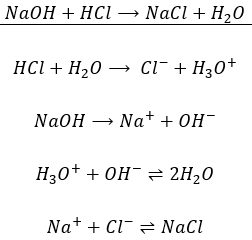

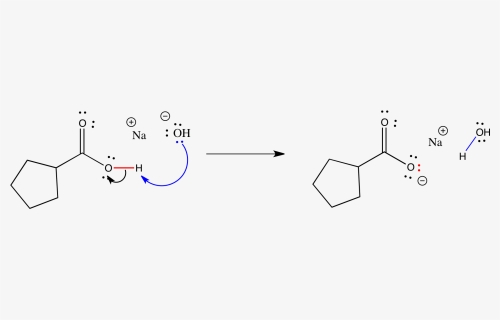
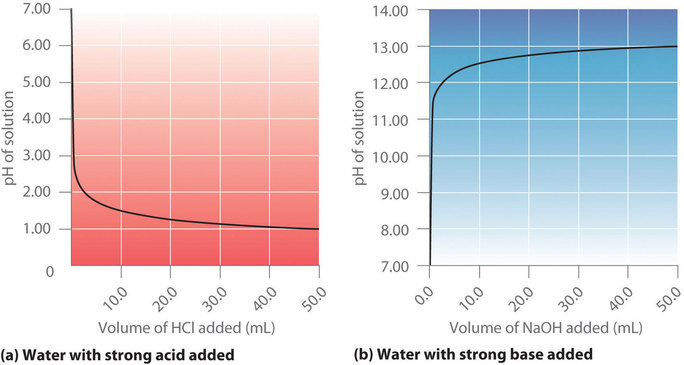
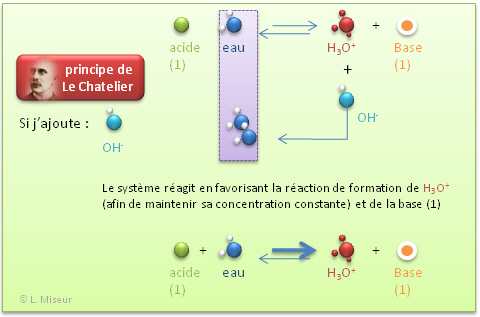





/prepare-sodium-hydroxide-or-naoh-solution-608150_FINAL-696b52d6f90b4b1383ec8f95db73a1f3.png)